The industrial calibration services market represents a $5.7B opportunity today, growing to $8.1B by 2030 at 5.3% CAGR. Despite being compliance-critical across aerospace, pharma, manufacturing, and energy sectors, the calibration procurement process remains fragmented, relationship-driven, and offline.
The opportunity: Build an AI-powered calibration intelligence platform that serves as the "single pane of glass" for enterprise calibration management—automating due-date tracking, lab matching, quote comparison, certificate management, and compliance reporting. Why this fits AIM.in: Calibration is the ultimate high-trust, high-compliance B2B service. Every manufacturing facility, pharmaceutical plant, and aerospace supplier needs it. The decision is complex (ISO 17025 accreditation, turnaround time, uncertainty levels, industry specialization) but the workflow is standardized—perfect for AI-driven matching.1.
Executive Summary
2.
Problem Statement
Who experiences this pain?
Spreadsheet Hell: Most companies track calibration due dates in Excel. When equipment is due, someone manually searches for service providers.
Quote Chaos: Getting quotes requires calling 3-5 labs, explaining equipment specs repeatedly, and waiting days for responses.
Certificate Management: Paper certificates get filed in binders. When an auditor asks for a calibration history, someone spends hours digging through physical records.
Compliance Anxiety: Missing a calibration due date can invalidate test results, trigger recalls, or fail audits. The stakes are high, but tracking is primitive.
Lab Selection Opacity: How do you know which lab can handle your Fluke 8846A multimeter calibration with 4-digit uncertainty? You call around and hope someone knows.
- Quality Managers at manufacturing plants tracking 500-5,000 pieces of equipment
- Metrology Lab Coordinators managing calibration schedules across multiple sites
- Compliance Officers preparing for FDA, AS9100, or ISO audits
- Procurement Teams negotiating with calibration service providers
3.
Current Solutions
| Company | What They Do | Why They're Not Solving It |
|---|---|---|
| Fluke Calibration | OEM calibration services + management software (MET/CAL) | Vendor lock-in; expensive software; focused on their equipment |
| Trescal | World's largest calibration service provider | Services company, not a marketplace; no price transparency |
| Keysight | OEM calibration for Keysight instruments | Limited to their brand; not a platform |
| Caliber | Calibration management software | Just tracking, no marketplace or procurement integration |
| Qualer | Asset management and calibration tracking | Better UX but still no intelligent matching or marketplace |
| IndySoft | Calibration management system | Desktop-focused; no AI; no lab network |
4.
Market Opportunity
- Market Size: $5.7 billion (2023), growing to $8.1 billion by 2030
- CAGR: 5.3% (2023-2030)
- Growth Drivers:
| Segment | Size | Key Requirements |
|---|---|---|
| Mechanical | ~30% | Pressure gauges, torque wrenches, dimensional |
| Electrical | ~25% | Multimeters, oscilloscopes, power supplies |
| Thermodynamic | ~20% | Temperature sensors, ovens, chambers |
| Dimensional | ~15% | CMMs, micrometers, calipers |
| Physical/Chemical | ~10% | Pipettes, pH meters, scales |
5.
Gaps in the Market
Gap 1: No Price Transparency
Calibration is quoted like auto repair—you don't know if $450 for a Fluke 87V calibration is fair until you call five labs. A marketplace with transparent pricing would be revolutionary.Gap 2: Accreditation Verification is Manual
ISO 17025 accreditation varies by scope. Lab A might be accredited for DC voltage but not AC. Verifying a lab can actually calibrate your specific equipment at your required uncertainty requires reading PDF scope documents. AI should handle this instantly.Gap 3: No Predictive Maintenance Integration
Modern equipment can self-report drift patterns. Calibration should be condition-based, not calendar-based. Nobody is connecting equipment telemetry to calibration scheduling.Gap 4: Certificate Management is Stone Age
Digital certificates exist but aren't standardized. There's no "certificate wallet" where all your calibration records are stored, searchable, and audit-ready.Gap 5: Multi-Site Coordination is Painful
Enterprises with 50 facilities manage calibration independently at each site. There's no aggregated view, no volume pricing optimization, no standardized vendor relationships.6.
AI Disruption Angle
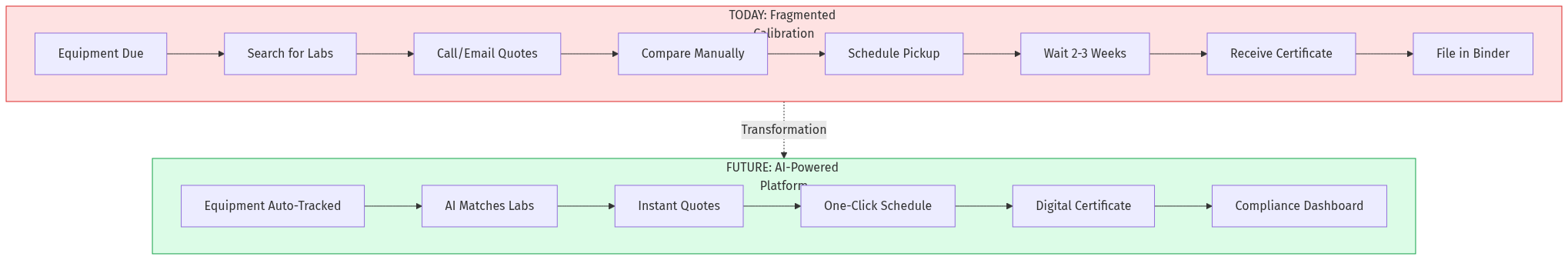
Intelligent Lab Matching
- Parse ISO 17025 scopes using NLP to match equipment to accredited labs
- Factor in turnaround time, historical quality scores, and geographic proximity
- Recommend labs based on similar equipment calibrated successfully
Predictive Due-Date Optimization
- Analyze equipment usage patterns to suggest calibration timing
- Account for environmental factors affecting drift
- Reduce over-calibration (waste) and under-calibration (risk)
Automated Quote Aggregation
- Standardize equipment specifications for consistent quoting
- Collect quotes from network labs in real-time
- Apply AI to predict realistic lead times and costs
Compliance Intelligence
- Auto-generate audit reports with one click
- Flag gaps in calibration coverage before auditors find them
- Track industry-specific requirements (AS9100, IATF 16949, FDA 21 CFR)
Certificate Digitization
- OCR legacy paper certificates into searchable database
- Standardize certificate format across all labs
- Create tamper-evident digital certificates with blockchain anchoring
7.
Product Concept

Core Platform Features
Asset Registry- Import equipment lists from ERP/CMMS systems
- Auto-detect calibration requirements from model numbers
- Track location, custody, and usage history
- Visual dashboard of upcoming calibrations
- AI-optimized scheduling to batch equipment by location
- Integration with logistics providers for pickup coordination
- Searchable network of ISO 17025 accredited labs
- Real-time pricing and availability
- Quality scores based on on-time delivery and rejection rates
- One-click RFQ to multiple qualified labs
- Standardized spec sheets auto-generated
- Side-by-side comparison with total cost (service + shipping + rush fees)
- All calibration certificates in one searchable location
- Automatic linking to equipment records
- Audit trail and access controls
- Real-time compliance score by site, equipment type, or regulation
- Gap analysis and remediation recommendations
- Exportable reports for auditors
8.
Development Plan
| Phase | Timeline | Deliverables |
|---|---|---|
| MVP | 12 weeks | Asset registry, due-date tracking, basic lab directory (100 labs), certificate upload |
| V1.0 | +8 weeks | Quote request workflow, lab matching algorithm, compliance reports |
| V1.5 | +8 weeks | Marketplace payments, expanded lab network (500+), mobile app |
| V2.0 | +12 weeks | Predictive calibration, IoT integration, blockchain certificates |
- Backend: Node.js + PostgreSQL
- AI: GPT-4 for spec parsing, custom ML for lab matching
- Integrations: SAP, Oracle, Maximo, Dynamics 365
- Mobile: React Native
9.
Go-To-Market Strategy
Phase 1: Vertical Focus (Pharma)
Pharmaceutical companies face the most intense calibration compliance pressure. FDA 483 observations for calibration failures are public record—target companies cited in the last 24 months. Outreach:Phase 2: Lab Network Build
The marketplace is only valuable with lab supply. Strategy:- Start with regional labs (50-100 employees) who need customer acquisition
- Offer free listing with commission-only model (10-15% of service value)
- Prioritize labs with broad accreditation scopes
Phase 3: Enterprise Land-and-Expand
- Enter through a single manufacturing site
- Prove ROI: time saved, compliance improvements, cost reduction
- Expand to other sites and equipment types
Phase 4: Channel Partnerships
- Partner with ERP vendors (SAP, Oracle) for embedded calibration management
- White-label platform for large calibration service providers
- Integration with CMMS platforms (Maximo, Fiix, UpKeep)
10.
Revenue Model
SaaS Subscription (Asset Management)
| Tier | Assets | Price | Features |
|---|---|---|---|
| Starter | 1-100 | $299/mo | Basic tracking, 50 lab directory |
| Professional | 101-500 | $799/mo | Full marketplace, compliance reports |
| Enterprise | 500+ | Custom | Multi-site, integrations, dedicated support |
Marketplace Commission
- 10-15% of calibration service value facilitated through platform
- Paid by lab (not customer)
- Expected average order: $350 → $35-52 commission
Premium Services
- Certificate digitization: $2 per document
- Rush matching: $50 per expedited request
- Compliance consulting: $500/audit prep session
| Year | Customers | ARR (SaaS) | GMV (Marketplace) | Commission | Total Revenue |
|---|---|---|---|---|---|
| Y1 | 50 | $300K | $2M | $200K | $500K |
| Y2 | 200 | $1.2M | $10M | $1M | $2.2M |
| Y3 | 500 | $3M | $30M | $3M | $6M |
11.
Data Moat Potential
Proprietary Data Assets:
Lab Performance Database
- On-time delivery rates
- Calibration rejection rates
- Turnaround time by equipment type
- Customer satisfaction scores
Equipment Intelligence
- Which models require frequent calibration
- Common failure modes and drift patterns
- Correlation between usage and calibration needs
Pricing Intelligence
- Fair market price for every equipment-service combination
- Regional pricing variations
- Volume discount patterns
Compliance Intelligence
- Industry-specific calibration requirements
- Regulatory change tracking
- Audit finding patterns and prevention
Network Effects:
- More labs → better matching → more customers → more data → better AI → more labs
- Lab quality data improves with transaction volume
- Enterprise customers attract other enterprises in same supply chains
12.
Why This Fits AIM Ecosystem
Perfect AIM Vertical:
Structured B2B Decision: Calibration is a complex decision with clear criteria (accreditation, uncertainty, turnaround, price) that can be structured into a decision engine.
High Trust Requirement: ISO 17025 accreditation and audit trails are mandatory. Platform trust signals (verified accreditation, quality scores, audit-ready records) become the core value prop.
Repeat Purchase: Every piece of equipment needs calibration every 6-24 months. Lifetime customer value is massive.
Domain Synergy: Could integrate with thefoundry.in (industrial procurement), testing-labs intelligence (existing article), and job-shop manufacturing.
India Opportunity: India's manufacturing boom (electronics, pharma, aerospace) creates massive calibration demand. Most calibration is still done by relationships with local shops—a marketplace can professionalize this.
Lab acquisition: Labs may resist joining a platform that commoditizes their services
Enterprise sales cycles: Quality departments move slowly; could take 12-18 months to close
Integration complexity: Every customer has different ERP/CMMS systems
Regulatory risk: FDA might mandate specific calibration vendors, limiting choice
## Mental Models Applied
ZEROTH PRINCIPLES: What are we assuming?
- Assumption questioned: "Calibration requires human expertise to match equipment to labs."
- Reality: ISO 17025 scopes are structured data. Equipment specifications are standardized. This is fundamentally a matching problem that AI can solve.
INCENTIVE MAPPING: Who profits from status quo?
- Incumbent labs: Large service providers like Trescal benefit from opacity—customers don't comparison shop
- OEMs: Fluke, Keysight prefer customers use their calibration services at premium prices
- Breaking it: A transparent marketplace disrupts both by forcing price competition
DISTANT DOMAIN IMPORT: What other field solved this?
- Healthcare credentialing: Platforms like Modio verify physician credentials across hospitals. Same pattern: verify accreditation, match to requirements, manage compliance
- Auto service: Openbay and RepairPal created transparent marketplaces for car repair. Calibration is "repair for measurement equipment"
FALSIFICATION (Pre-Mortem): Why would this fail?
STEELMANNING: Best argument against
- "Relationships matter. My calibration lab knows my equipment history, comes on-site, and fixes problems fast. A marketplace can't replicate that."
- Counter: We don't replace relationships—we enable informed relationships. Small labs gain access to customers they'd never reach. Customers gain transparency. The best labs win.
## Verdict
Opportunity Score: 8.5/10 Strengths:- Large market ($8B) with proven growth
- Compliance-critical = sticky customers
- Fragmented supply = marketplace opportunity
- AI can genuinely add value (matching, prediction, automation)
- Strong data moat potential
- Recurring revenue from repeat calibration cycles
- Long enterprise sales cycles
- Lab network chicken-and-egg problem
- Integration complexity with legacy systems
- Regulatory changes could disrupt market
## Sources
❧